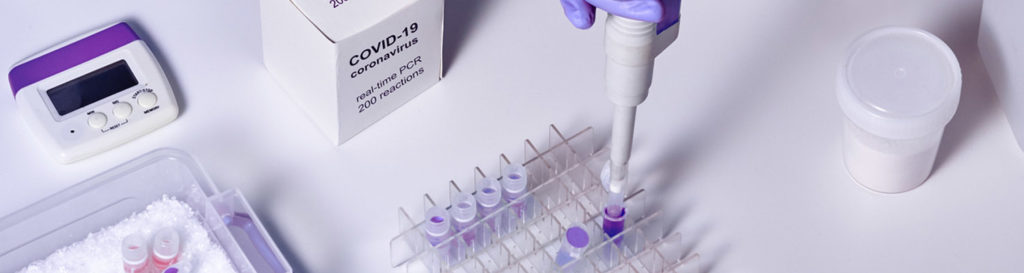
COVID-19 Detection Kit- Extraction system and PCR system
- The CoV & Flu A/B Detection Kit is a one-step real-time reverse transcription polymerase chain reaction (RT-PCR) test for qualitative detection of SARS-CoV-2 and Flu A/B specific RNA.
- CoV & Flu A/B Detection Kit is for use only by laboratories certified to perform high complexity tests and is limited to clinical laboratory personnel specifically instructed and trained in the techniques of real-time PCR and in vitro diagnostic procedures.
- This extraction kit has to be used in conjunction with reagent (Extraction & Purification Reagents)
- CE certified, FDA approval under process

Extraction & Purification Reagents
- Extraction & Purification Reagents is for extracting and purifying nucleic acids from specimens by using proteinase K, magnetic beads and buffering solutions.
- The purified nucleic acids can be directly used for downstream testing. This kit should be used with Auto Extraction Instrument- CoV & Flu A/B Detection Kit. If hospital have Extraction kit already, then use only reagent.
- The Extraction & Purification Reagents contains Reagent Pre-filled Plate, Proteinase K and Magnetic Rob 8-cover Strips.
- This kit uses a unique type of magnetic beads and buffer system to isolate and purify high quality nucleic acid from serum, plasma, swab, urine and other clinical samples. The magnetic beads coated with silica gel have a strong affinity to nucleic acids under certain conditions. When conditions change, magnetic beads can release nucleic acids, thereby achieving the purpose of rapid isolation and purification of nucleic acids.
- CE certified, FDA approval under process
Nucleic Acid Isolation Machines
- It’s an advance and automated machine for the Automated Nucleic Acid Extraction (RNA) and Automated processing to save labors and improve efficiency and much safer and accurate in terms of giving results.
- 3 RNA Extraction + 1 qPCR System + 3-4 operators = Nearly 1000 tests/12 hours.
- This machine is efficient enough to distinguish 2019-nCoV from flu viruses.
- CE certified, FDA approval under process
CORONAVIRUS Rapid Testing Kit
This is self-testing kit and result is available in 10 minutes.
- 2019-nCoV antibody detection reagent. This reagent is used for the qualitative detection of 2019-nCoV antibodies in human serum, plasma and venous whole blood samples in vitro, and is suitable for the preliminary screening of clinical 2019-nCoV infection.
- Test result available in 10 minutes at the conform of our home.
- It is CE certified and FDA approved.
To know more about these products Please contact "COVID19.Support@dia33.com" or CLICK HERE.

